Fiber Photometry Solutions
Choose the right fiber photometry system for YOUR experimental needs.

All-in-one solutions
- Acquisition -> Data analysis
- Combine with behavior
- Compatible with optogenetics
- Plug-and-play
Fiber Photometry Systems
Fiber photometry is a neuroimaging technique that monitors neuronal activity in freely-moving animals. This technique utilises genetically encoded fluorescent indicators (e.g. GCaMP, dLight, GRAB-Ach, RCaMP, jRGECO1) expressed in target brain regions. These indicators fluorescence only when bound to melocules such as calcium, dopamine or acetylcholine, reporting real-time molecular dynamics during complex behaviors.
Unlike Doric Miniscopes, which resolve single-cell activity, fiber photometry records population signals (i.e bulk fluorescence from labelled neurons). But unlike microscopy, fiber photometry is minimally invasive, optimized for multi-animal & multi-site experiments, and more cost-effective.

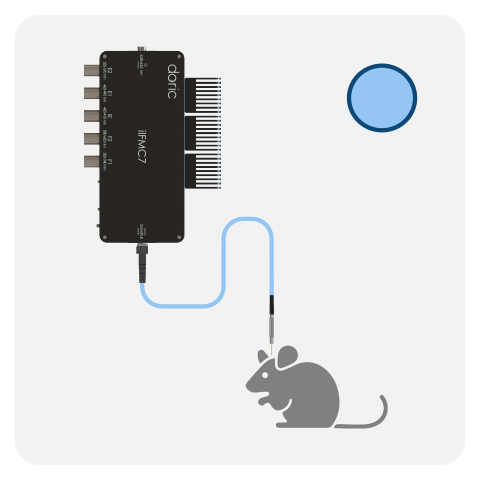
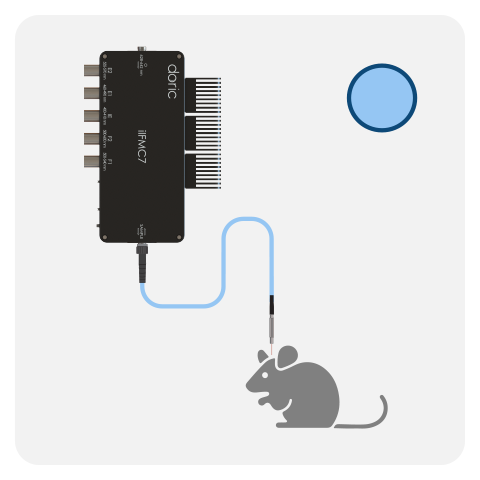
Fiber photometry system are composes of several core components that, together, combine multiple wavelengths into a single or mutliple fiber(s) and collect the emitted fluorescence response, including:
- Console
- LED Driver & LEDs
- Fluorescence mini cube (FMC)
- Fiber-optic patch cords
- Rotary Joints (commutators)
- Fiber-optic cannulas
- Data Acquisition & Analysis Softwares
The latest generations of Doric systems integrate multiple components into a single device, providing several advantages. Learn more.
Browse below the role of each fiber photometry component:
Doric Lenses Inc. is a recognized leader in advanced fiber photometry solutions for behaving animals, driving innovation in this rapidly evolving field. Each system was carefully engineered to address the specific experimental needs & challenges.
The three main types of Fibers Photometry Systems are split into categories based on the type of detector.
Explore how Doric’s fiber photometry systems compare across key metrics.
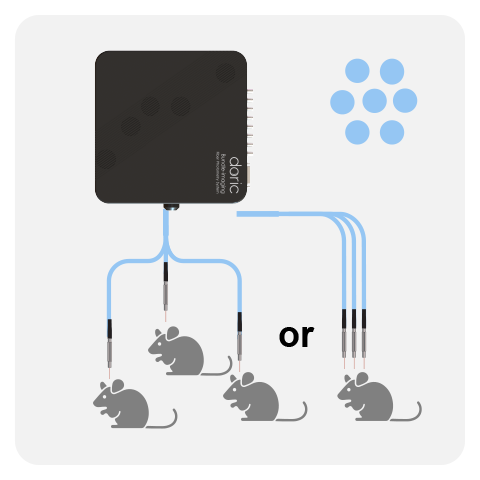
All Basic Fiber Photometry Systems are fitted with a high-sensitivity Doric photodetector. In the latest generations, the photodetector is integrated directly within the fluorescence mini cube / headstage (FMC, RFMC and WiFP), providing higher signal-to-noise ratio compared to non-integrated version. All Basic systems can be run in either lock-in mode (frequency-based division) or interleaved mode (time-based division) using live demodulation/deinterleave algorithms in (free) Doric Neuroscience Studio software.
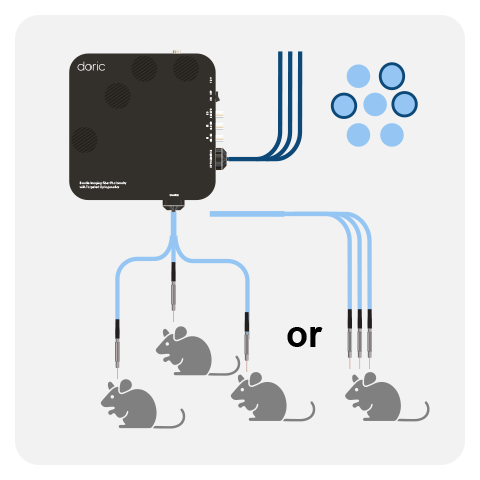
One FMC/headstage can record the GFP-based and/or RFP-based signal from a single optical fiber. However, it is possible to combine multiple cubes/headstages to record from more than one mouse or brain site (up to 4-8*) when using the Neuroscience Console 500.
BASIC
The latest generation of Basic Fluorescence Mini Cube (FMC) were disigned for flexible isosbestic point, such that the integrated isosbestic LED can be swapped between: 405, 415, 425 and 440 nm LEDs. In addition, both the 1-color and 2-color configuraitons are compatible with simultaneous red-shifted optogenetics, without crosstalk. The Basic system can be combined with pigtailed rotary joints (FRJ_1x1_PT, FRJ_2x2_PT & AFRJ_2x2_PT) for long-term, freely-moving experiments. With decade-long validation, the basic fiber photometry system is ideal to record GFP, RFP and isosbestic signals from 1-2 sites/animals, with the possibility to expand later on.
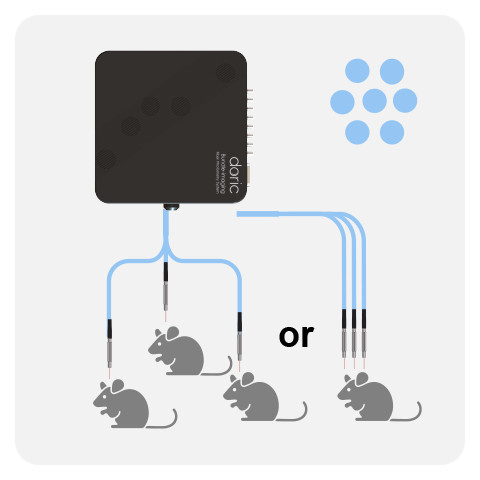
ROTARY BASIC
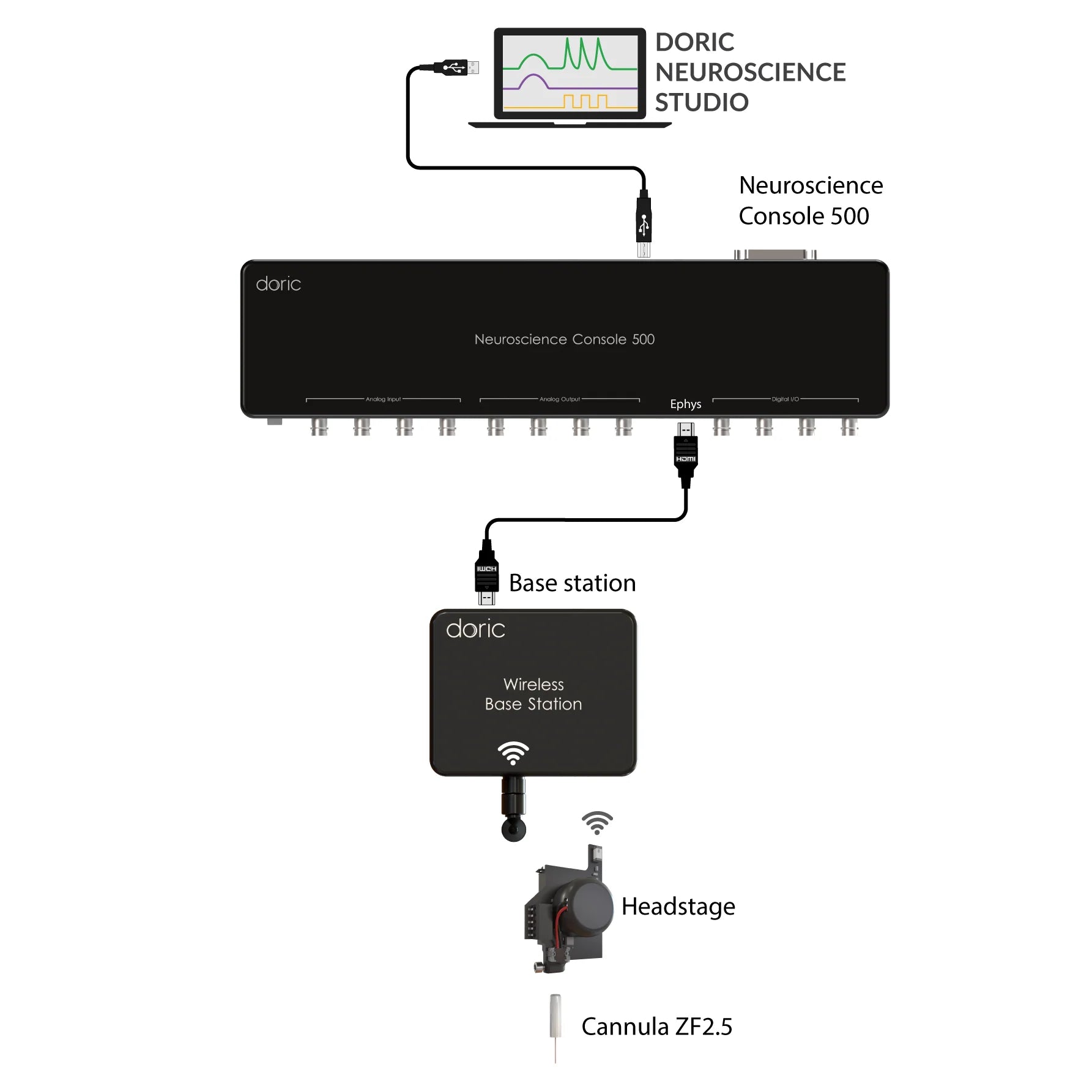
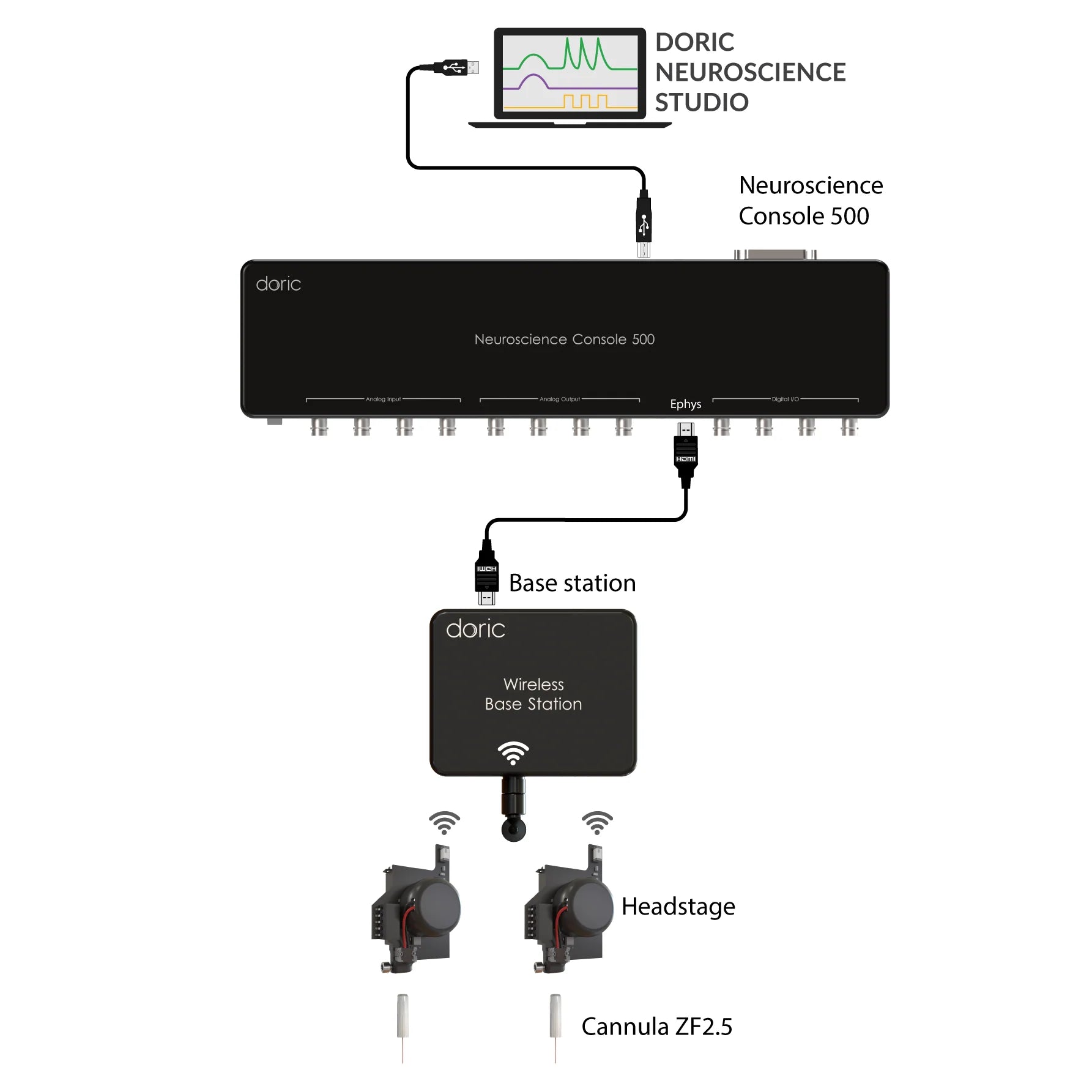
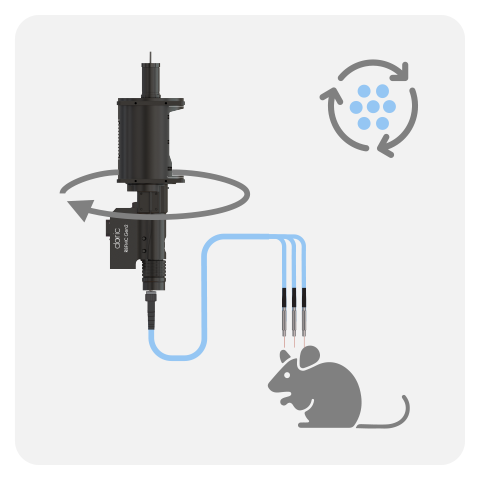
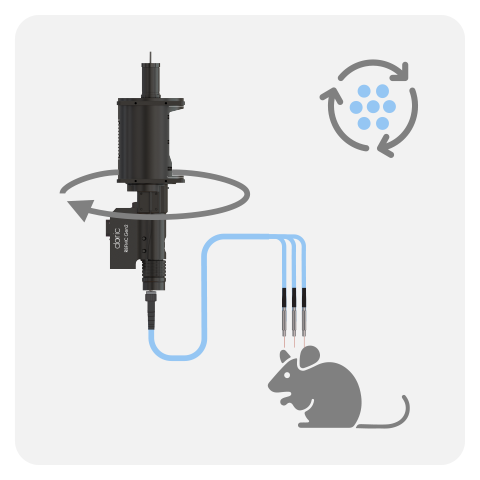
In Rotary Fiber Photometry Mini-Cube (RFMC), up to two fluorescence mini cube(s) are built-on the rotary joints itself. This unique design abolishes rotational artifacts in the signal and minimizes the number of connections before the photodetector. Therefore, RFMC system boasts a higher quality signal compared to basic FMC system paired with an external rotary joint, and is ideal for experiments lasting several hours or even days. The rotary joint also contains a hollow channel that can be used for fluid delivery, electrophysiology/EEG, or optogenetics.
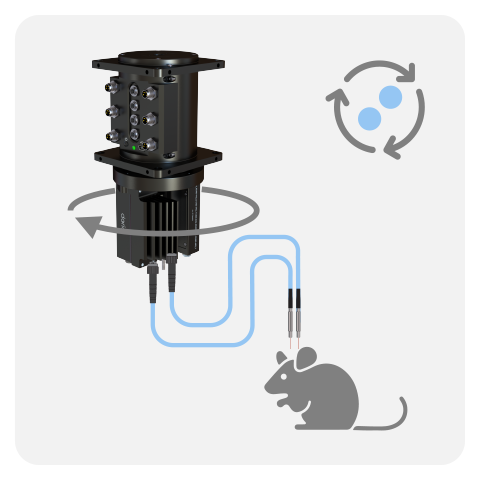
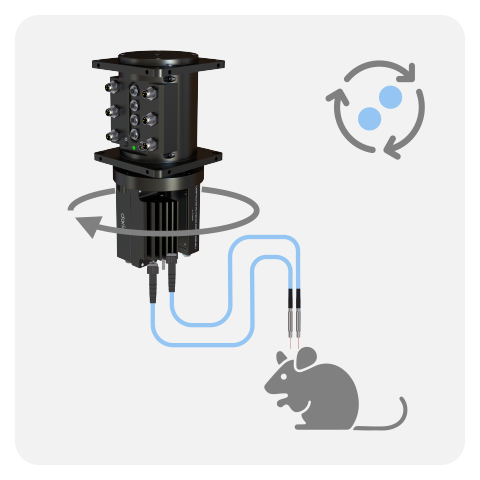
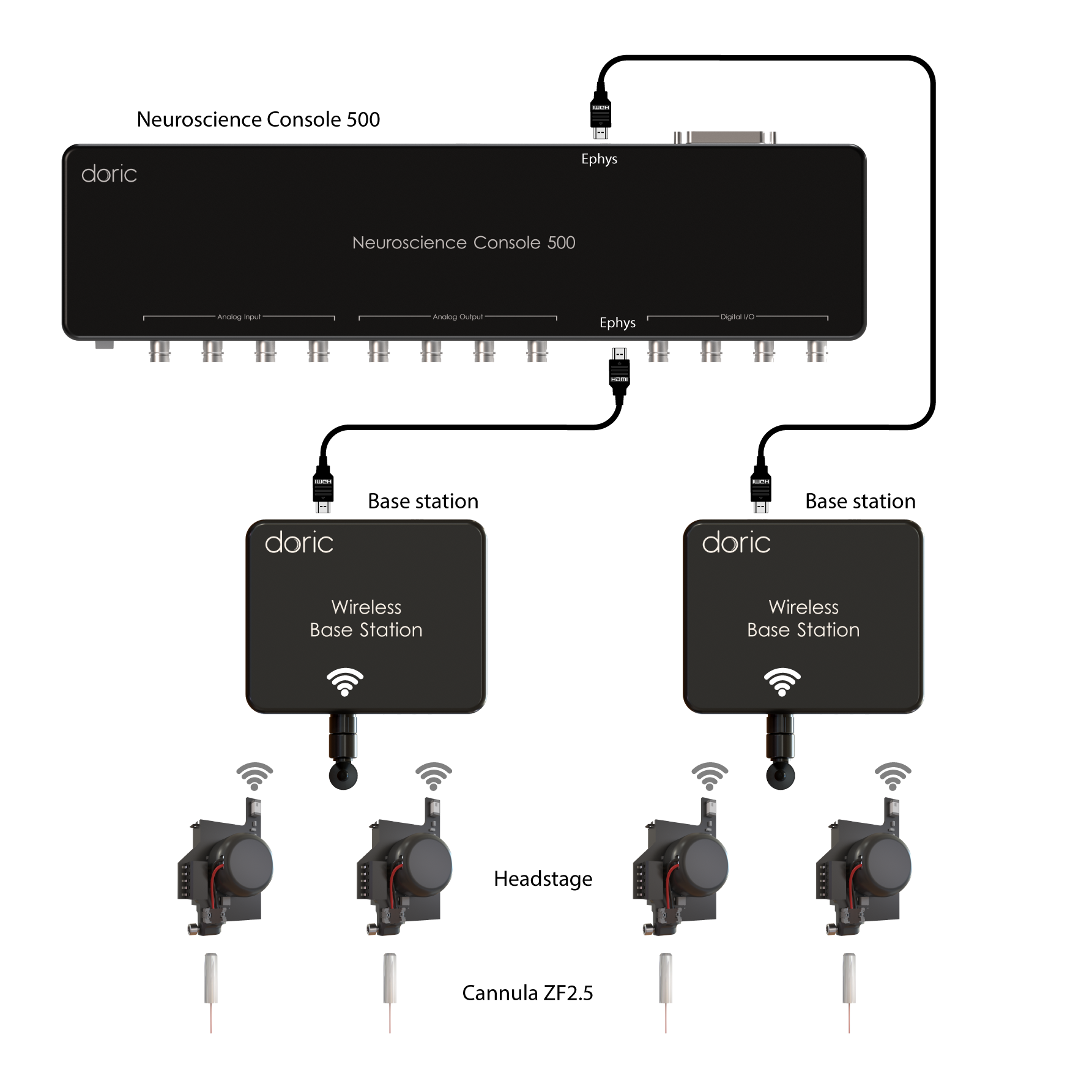
WIRELESS
Lastly, the Wireless Fiber Photometry System (WiFP) was designed to circumvent tangling issues when recording multiple animals in the same cage or for behaviors disrupted by tethered system. The ilFMC4 (with 470 and isosbestic LED and detector) were miniturized onto a lightweight headstage. Multiple headstages can be run at the same time, thus this system is ideal to study neural population underpinning social behaviors.
* Depending on the type of system and wether its 1-colour or 2-color configuration:
- Basic: 1-color (up to 8 sites/animals) and 2-colors (up to 4 sites/animals);
- Rotary: Max 4 animals (max 1-2 sites per animal, 1- or 2-colors);
- Wireless: Max 4 animals (1-site, 1-color per animal) .
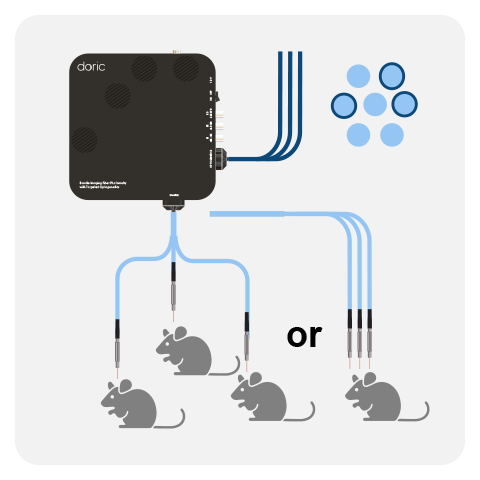
Unlike the Basic Fiber Photometry Systems, which require one mini cube per site/animal, the Bundle-imaging Fiber Photometry systems use a CMOS camera to image the entire fiber bundle simultaneously. Bundle-Imaging Fiber Photometry System are ideal when recording 1- or 2-colors from multiple animals or brain regions (1-19 fibers), all at a reasonable price.
All bundle-imaging system are available with 2-color configurations (plus isosbestic) and the three excitations are interleaved in time (i.e the lock-in mode is not available). All three LEDs excite the entire fiber bundle (no independent power adjustement), and the photometry signal is then sampled at 20-100 Hz depending on the field-of-view of the camera and the number of fibers imaged at the same time.
Three bundle-imaging system options differ based on the level of integration and compatibility with optogenetics:
BUNDLE-IMAGING (GEN.3)
The BFMC-G3 is fully integrated, plug & play design (with CMOS camera, LEDs, LED Driver & console in a single device), greatly simplifying the set-up & troubleshooting. However, this system is not compatible with simultaneous optogenetics.
BUNDLE-IMAGING WITH TARGETED OPTOGENETICS (BFTO)
The BFTO system is specifically designed for multi-fiber photometry with targeted optogenetics. The term "targeted" refers to independent, multi-site optogenetic control is ideal for close-loop optogenetics (both multi-animal and/or multi-site) experiments. This system provides the greatest flexibility for common neuroscience experiments.
ROTARY BUNDLE-IMAGING
The RBFMC is designed for single-animal, multi-site experiment lasting several hours/days. The CMOS detector is integrated directly on the rotating joint ABOLISHING rotation-related artifacts, allowing for high-quality signals. The 2025 version now comes with a flexible isosbestic design, easily switching between different biosensors. Additionally, the RBFMC includes optogenetic capabilities that can illuminate all sites simultaneously* or can be fitting with external laser diode for independent optogenetic stimulation on a non-fiber photometry site.
* Simultaneous optogenetic on the fiber photomery sites is only available for the 1-color configuration; For 2-color configuration optogenetics can be added, but on a non-photometry site
The latest innovation in fiber photometry!
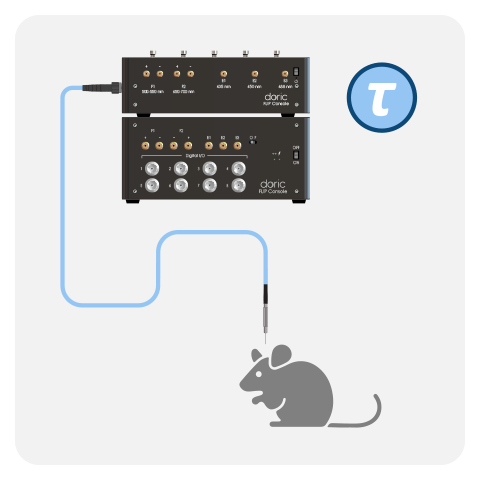
Fluorescence lifetime fiber photometry measures changes in the fluorescence decay time of indicators rather than signal intensity, providing a readout that is largely independent of motion artifacts, photobleaching, and fiber coupling fluctuations. This makes it especially powerful for stable, tonic or baseline changes in neural activity or biochemical states in freely-behaving animals.
WHAT IS FLUORESCENCE LIFETIME?
Fluorescence lifetime is the average time a fluorophore remains in its excited state before emitting a photon, typically on the nanosecond timescale. It is an intrinsic property of the fluorophore that depends on its local molecular environment rather than its concentration or excitation intensity.

Want to learn more?
Register for the Discover FluoPulse webinar, run quartely (or request a recording).
SYSTEM COMPARISON
This side-by-side comparison highlights differences in channel configuration, maximum number of site/animal(s), dual-color & optogenetics compatibility, and advanced features across seven distinct fiber photometry systems.
| BASIC | BUNDLE-IMAGING | LIFETIME | ||||||
|---|---|---|---|---|---|---|---|---|
|
|
|
|
|
|
|
|
||
| Fiber Photometry |
Flexible isosbestic | ✔ | ✔ | n/a | ||||
|
Max # sites
●
◐
|
Up to 8 Up to 4 |
1–2 | 1 ✖ |
Up to 9–19* | Up to 9–19* | Up to 7–19* | 1–2 1 |
|
|
Multi-animal (separate cage)
●
◐
|
Up to 8 Up to 4 |
Up to 4 ✖ |
Up to 9–19* | Up to 9–19* | 1–2 ✖ |
|||
|
Multi-animal (same cage)
●
|
Up to 4 | |||||||
| Optogenetics | Same site as photometry | ✔ | ✔ | ✔*** | ✔ | |||
| Different site as photometry | ✔ | ✔ | ✔ | ✔ | ✔ | ✔ | ||
| Behavior (Rotary joint / commutator) |
Long-term, freely-moving behavior | ** | ✔ | 2–4 h | ** | ** | ✔ | ✔ |
| Integrated rotary joint | ✔ | n/a | ✔ | n/a | ||||
| Interacting animals | ✔ | |||||||
| Add Ephys / EEG & EMG | ** | ✔ | ** | ** | ✔ | ** | ||